Chemistry is the study of compositions, properties and behaviors of matter in the universe. Chemists look at atoms and their interactions with other atoms to form chemical compounds. Chemistry has been referred to as the central science as it bridges natural sciences with each other.
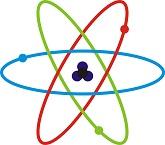
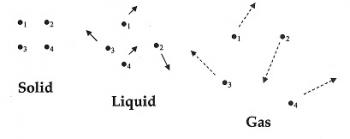
The current and most widely accepted model of atomic structure is the quantum mechanical model. Matter can be studied in a solid, liquid or gaseous state. It can also be studied in either an isolated system or in a combination of phases and species. The interactions of the atoms in these phases and species will typically result in a reaction or transformation. These interactions can create a useful substance often utilized by industries.
A chemical transformation of a reaction can be symbolized by a chemical equation. This shows the transformation from one chemical compound to another. In a chemical equation, the number of atoms on the left must equation the number of atoms on the right. This follows the principle that matter cannot be created or destroyed.
A chemical element is a substance which is made of a single atom type. The type of atom is determined by the number or protons in the nuclei. The number of protons determines the atomic number. The mass of the element is determined by the sum of protons and neutrons present in the nucleus.
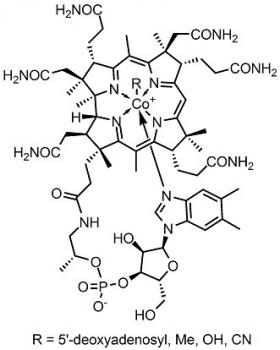
A compound is composed of more than one element. An example of a compound is carbon dioxide. A molecule is many atoms bound together by covalent bonds. In a molecule all valence electrons are paired with other electrons. Due to this the molecule is electrically neutral.
Chemistry is being utilities in many aspects of everyday life. Everything that you come in contact with uses chemistry principles. The chemical industry is a very important industry with respect to economics. In 2004, the chemical industry grossed over 587 billion dollars US.
© BrainMass Inc. brainmass.com May 7, 2026, 4:59 pm ad1c9bdddf