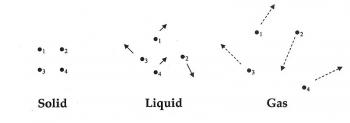
"The second law of thermodynamics states that the entropy of an isolated system never decreases due to the fact that isolated systems spontaneously evolve towards thermodynamic equilibrium"¹. Thermodynamic equilibrium is the state of maximum entropy.
The second law of thermodynamics can be understood and explained using the underlying quantum statistical mechanics with the assumption of low-entropy initial conditions in the distance past. In statistical mechanics, entropy is the measure of the number of microscopic configurations corresponding to a macroscopic state.
The second law of thermodynamics has been expressed in many ways. The following at the most predominantly used.
Clausius Statement
Heat can never pass from a colder to a warmer body without some other change.
Kelvin Statement
It is impossible to cool matter in a system below the temperature of the coolest of the surround objects
Principle of Caratheodory
In every neighborhood of any state S of an adiabatically isolated system there are states inaccessible from S.
References
1. Planck, Max. "Vom Relativen Zum Absoluten."Die Naturwissenschaften 13.3 (1925): 463-64. Print.
© BrainMass Inc. brainmass.com July 26, 2024, 12:29 pm ad1c9bdddf