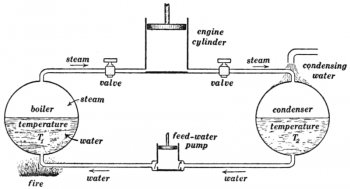
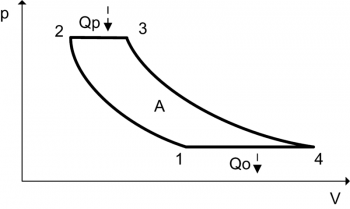
A heat engine is a system that converts heat to mechanical work. It does this by bringing a fluid from a higher temperature to a lower temperature. A heat source will create thermal energy that brings the fluid to the high temperature. The fluid creates work in the engine while transferring heat to the colder sink until it reaches a low temperature state. During this process some of the thermal energy is converted into work by exploiting the properties of the working substance. The fluid can be any system with a non-zero heat capacity.
Heat engines are different from other engine types by the fact that the efficiency is defined by the Carnot’s theorem. Although the efficiency restriction is a negative, an advantage of heat engines is that most energy forms can be converted easily to heat. Since the heat source supplies energy to the engine, it can be powered by virtually any kind of energy. Heat engines have a wide range of applications. Some examples of heat engines include steam engines, diesel engines and gasoline engines in a car. These are powered by the expansion of heated gases.
© BrainMass Inc. brainmass.com June 30, 2024, 9:22 am ad1c9bdddf