The last step of cellular respiration is Oxidative Phosphorylation (OXPHOS) using Electron flow or Electron Transport Chain (ETC) in the inner mitochondrial membrane. The term ‘oxidative’ refers to oxidation reactions, where electrons are transported to oxygen, and ‘phosphorylation’ denotes the synthesis of ATP. This process produces the largest amount of ATP, with a net gain of 34 ATP units out of the total of 38 ATP in cellular respiration.
The products of the Krebs cycle or Citric Acid Cycle are 6 NADH, 2 FADH2 and 2 ATP and waste products of CO2 and H2O. Namely, it is NADH and FADH2 that are reduced to release high-energy electrons to oxygen, an electron acceptor. These redox reactions take place in the electron transport chain, which is a series of protein complexes embedded in cristae membrane. Another function of the ETC is to couple redox reactions with the transport of protons (H+ ions) across the membrane, creating an electrochemical proton gradient or electron potential.1 Due to the established gradient, the electrons then flow down the chain, moving from electron donors to electron acceptors whilst creating ATP.
A number of key enzymes are used in Oxidative Phosphorylation, one of which is ATP synthase. ATP synthase undergoes chemiosmosis, which converts the energy gained from proton transfer to phosphorylate adenosine diphosphate (ADP) to adenosine triphosphate (ATP).
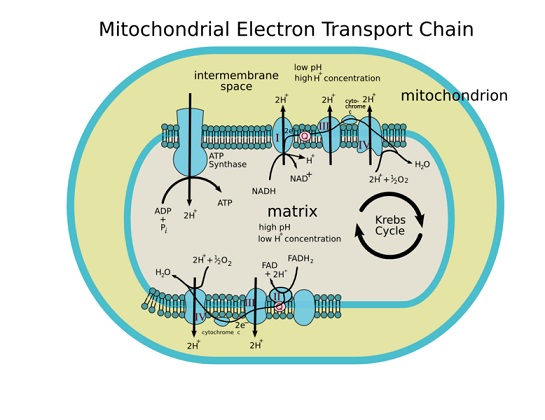
Here is a diagramatic outline of the Electron Transport Chain in the mitochondrial cristae.
References
1. Berg, J.M., Tymoczko, J.L., Stryer, L. (2002) Oxidative Phosphorylation, Chapter 18, Biochemistry, 5th Edition. Retrieved at http://www.ncbi.nlm.nih.gov/books/NBK21208/.
© BrainMass Inc. brainmass.com July 26, 2024, 4:33 am ad1c9bdddf